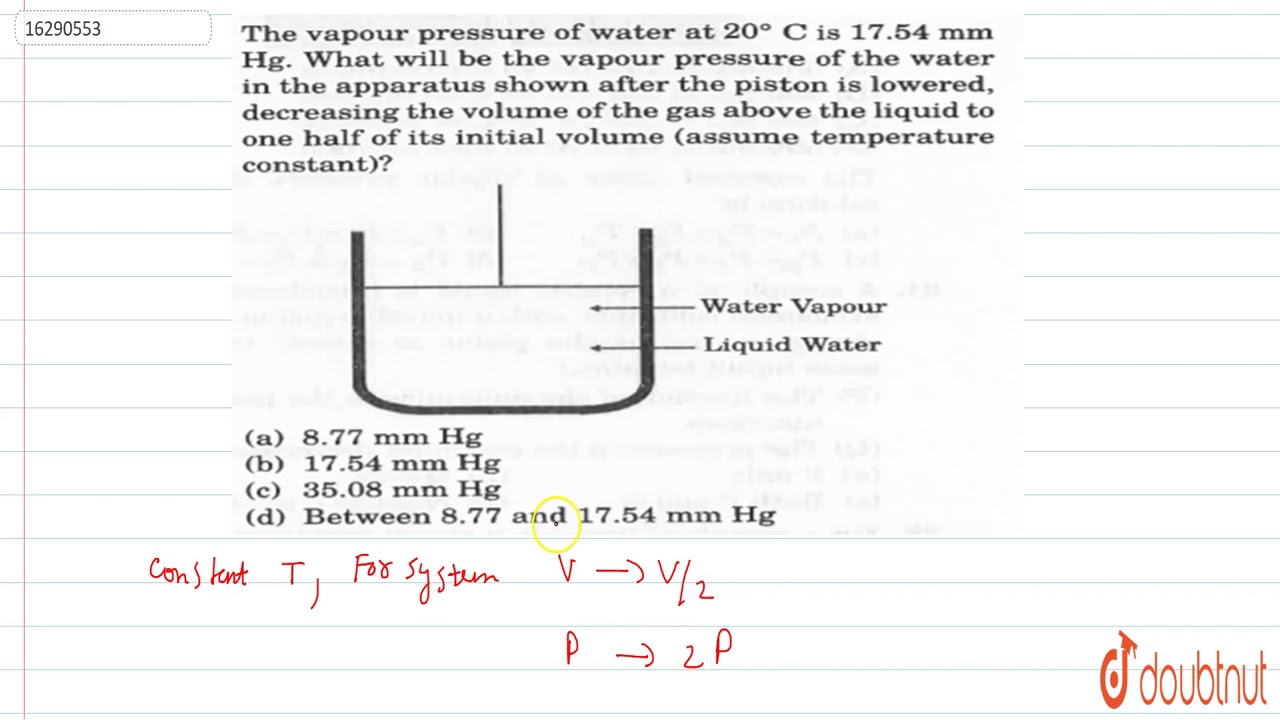
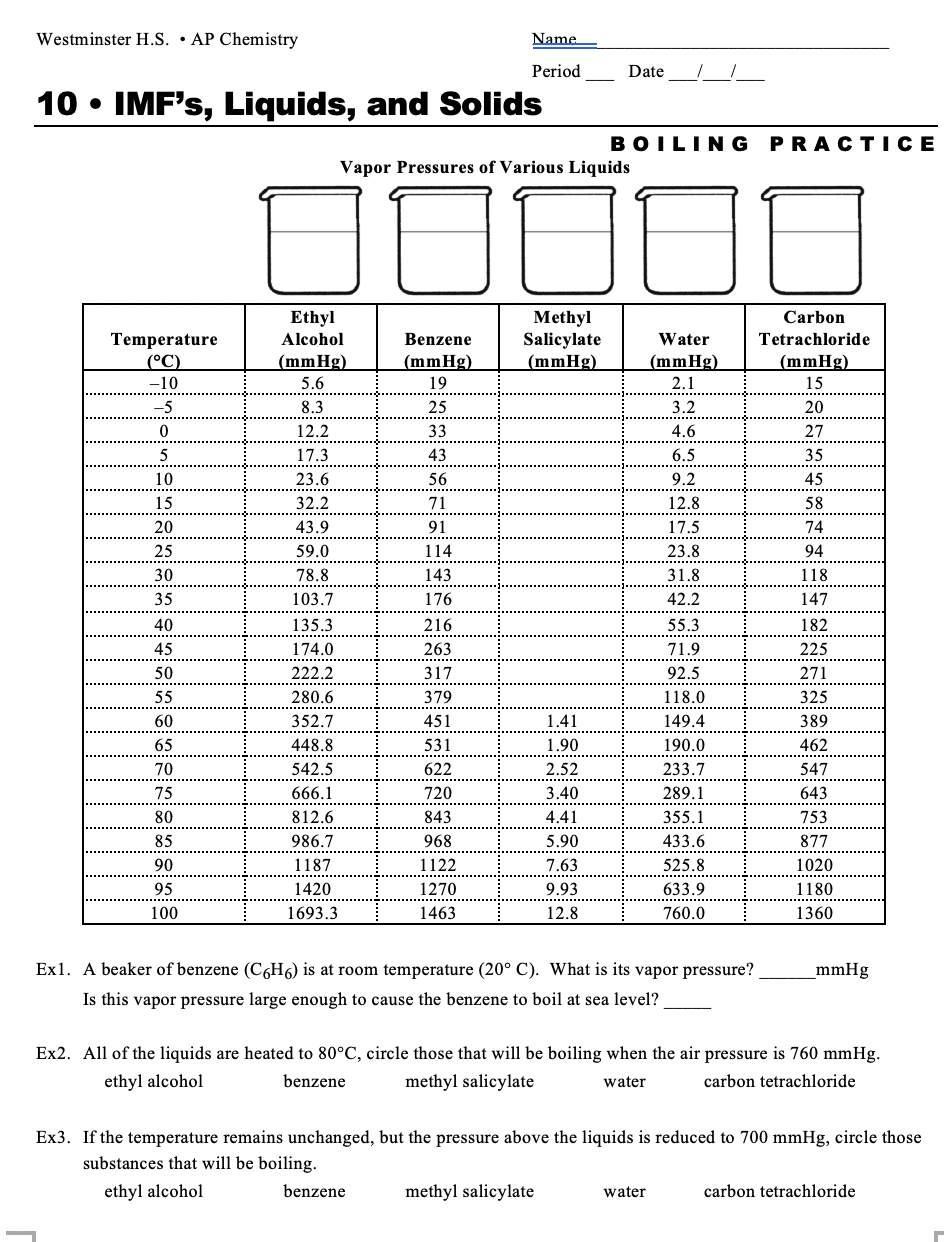
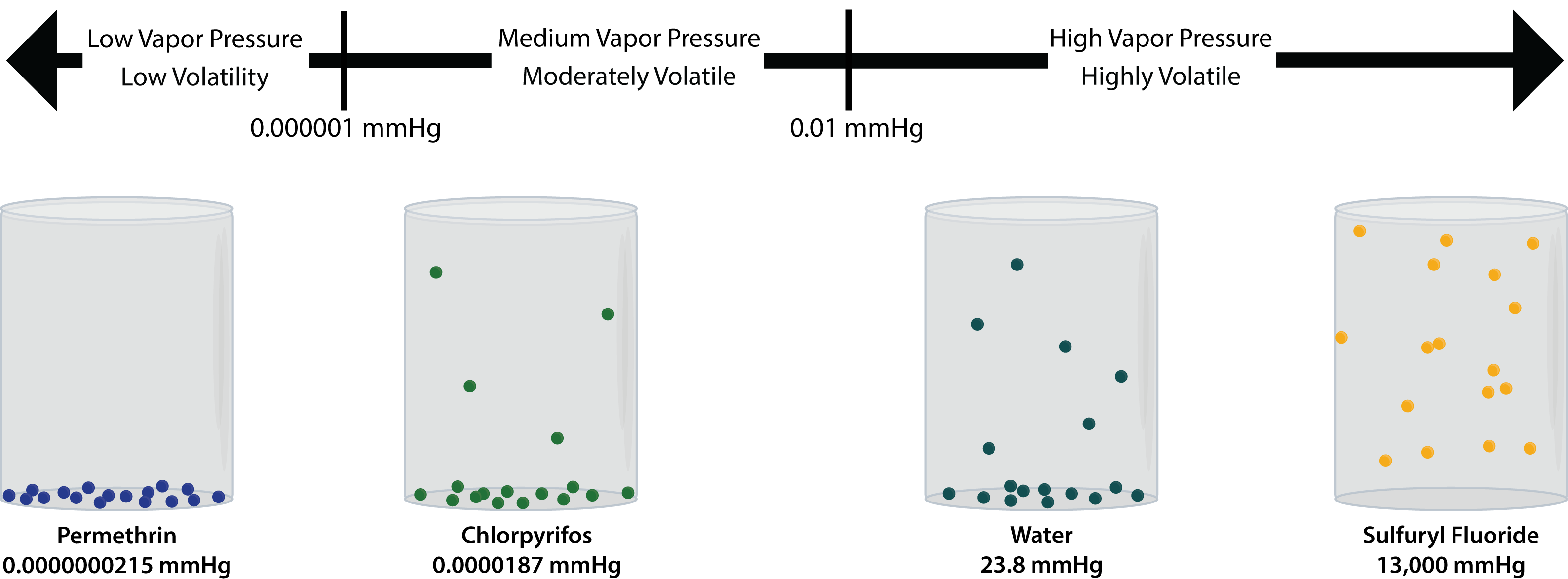
The vapour pressure of water at room temperature is 23 8 mm of hg the vapour pressure of an aqueous solution of sucrose with mole fraction 0 1 is equal to chemistry solutions.
The vapour pressure of water at room temperature is 23 8 mm hg.
Check answer and solution for above chemistry question tardigrade.
The vapour pressure of an aqueous solution of sucrose with mole fraction 0 1 is equal to.
Now from raoult s law we have.
The vapour pressure of an aqueous solution of sucrose with mole fraction 0 1 isequal to a 2 39 mm hg b 2 42 mm hg c 21 42 mm hg d 21 44 mm hg.
Weight of urea taken w 2 50 g.
The vapour pressure of water at room temperature is 23 8 mm hg.
The vapour pressure of an aqueous solution of sucrose with mole fraction 0 1 is equal to 3 a 23 9 mm hg b 24 2 hg c 21 42 mm hg d 21 44 mm hg.
The vapour pressure of water at room temperature is 23 8 mm hg.
We take vapour pressure as p 1.
It is given that vapour pressure of water p i o 23 8 mm of hg.
Now we have to calculate vapour pressure of water in the solution.
The vapour pressure of an aqueous solution of sucrose with mole fraction 0 1 is equal to.
The vapour pressure of wate.
Molecular weight of water m 1 18 g mol 1.
The vapour pressure of water at room temperature is 23 8 mm hg.
The vapour pressure of water at room temperature is 23 8 mm hg.
Molecular weight of urea m 2 60 g mol 1.
The vapour pressure of an aqueous solution of sucrose with mole fraction 0 1 is equal to.